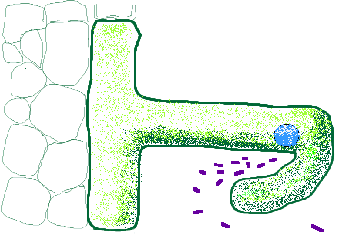
inf
The interaction of the Rhizobium bacteria in soil and the legume plant root is an extremely complex one, leading to drastic biological, morphological and biochemical changes in the plant root and the bacterial cells.
The various stages have been named and described in detail for many different plants. They have many features in common, but differ in some details.
Development of nodules (generalized for rhizobia)
Stages:
| roc | multiplication of rhizobia at or near the root surface |
| roa | adhesion of rhizobia to root hair surface |
| hab and hac | root hair branching and root curling |
| inf | formation of an infection thread |
| noi | nodule initiation: formation of nodule
meristem, nodule development and differentiation |
| bar | bacteroid release from infection thread |
| bad | bacterial differentiation |
| nif | onset of nitrogen fixation |
| cof | biochemical and physiological functions
associated with nitrogen fixation |
| nop | maintenance (persistence) of nodule function |
Diagrams of the process:
roc;
Rhizobia can live saprophytically within the soil. To colonize the
root and produce nodules they must migrate to the root surface. Two possible
mechanisms; chemotaxis-movement along a concentration gradient of a chemical
and electrotaxis-movement along electric currents flowing into actively
growing parts of the root and out of the older parts - rhizobia move along
the inwardly flowing electric current. Plant microbial interactions occur
before roc stage occurs. Root exudates can stimulate growth and movement
- homoserine is released by pea plants and is a preferred C and N nutrient
for R. leguminosarum biovar vivieae (for pea plant).
The rhizobial genes for nodulation (nod) are switched on by the components of the plant cell exudates. One such a component is luteolin (5,7,3",4" tetrahydroxyflavone). This is not specific and is produced by many plants. Some flavones actually inhibit nod genes. There are many compounds that inhibit and induce nod genes and many are flavones, isoflavones and related compounds.
The nod gene is thought to be complex; the nod ABC group seem to be present in all rhizobia examined. NodC codes for a protein that becomes incorporated into the bacterial surface membrane so that clusters of cysteine-rich amino acids protrude from the surface. NodA and nodB may produce low molecular weight plant growth promoting compounds that stimulate cell division; nodC may help in moving these bacteria to the plant.
Host plant cell division is often stimulated before the next stage (roa)
takes place. Some diffusible compounds or growth regulators may enter the
plant root and promote division. The rhizobia may also stimulate the plant
cells to produce growth regulating compounds.
roa;
The cells attach to the root hair of the host plant. This performed proceeds in stages with increasing levels of specificity and decreasing levels of reversibility. Most studies have been with soybeans and clover. The rhizobia attach by polar attachment in these plants and 2 substances are closely involved in recognition and attachment. The plant has LECTINSand the rhizobia have surface polysaccharides. The lectins (non-catalytic proteins produced by the host plant) aid in recognizing particular carbohydrate residues in rhizobia. The was confirmed elegantly by using a plant pathogenic bacterium Agrobacterium rhizogenes to carry the gene for pea lectin to a clover root. This root could then be nodulated by a pea rhizobial strain.
There are many other factors, most poorly understood, that are part
of the attachment process - fimbriae, cellulose fibrils and Ca2+-binding
proteins.
hac and hab
This is the stage of hair curling and branching. The nod ABC and nod D sequences are involved and modify the hormone status of the host plant root invoking hair curling and sometime branching.

inf

Infection may be through the root hair or through wounds or cracks between cells. Hair infection is the most common in crop plants. There is digestion of the cell walls of the root hair caused by some interaction between the plant and the rhizobia. It was thought that this involved pectic enzymes and other plant produced digestion enzymes, but the real process is much more complex. It is spatially confined within the root hair curled region and closely associated with the hair metabolism as is indicated by the movement of an enlarged nucleus close to the point of rhizobial entry. There are strong protoplasmic streaming movements in the root hair cells.
Once the hair cell wall is penetrated, a tubular structure called the infection thread is formed. It and consists of newly synthesized cell wall material surrounding the rhizobia. The centre of the tube is a glycoprotein containing some bacterial products and some host glycoproteins. The whole structure is surrounded by a membrane contiguous with root hair plasmalemma.
noi

Some nodule formation processes infect through wounds or cracks in the
surface and a few can penetrate through direct epidermal penetration.

Nodule initiation depends on cell division (this maybe initiated earlier than the infection or noi stage). Cortical divisions precede infection. If the divisions are to proceed to form an organized nodule, then some of the daughter cells must become infected with rhizobia and some must differentiate into other specialized cell types. There are three types of such infections. Each cell may become infected by a branch of the infection thread. Some cells remain uninfected (pea). The second type is where only a limited number of cells
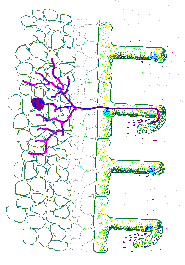
become infected and then enlarge to form nodules of restricted size
(soybean). The third type is where an undifferentiated zoogloea of cells
infect the root cells (not an infection structure).
The interstitial cells are not essential to some systems they play an essential metabolic role in others.
The intercellular spaces are sometimes filled with a glycoprotein similar to gum arabic and it may play a role in restricting oxygen diffusion. The peripheral vascular system forms to supply the nodule with nutrients and remove products.
nif

In pea and soybean, nitrogenase synthesis follows closely the release of bacteria from the infection thread. Other plants do not release the bacteria but still form nitrogenase. Haemoglobin is formed at about the same time. Haemoglobin acts to maintain a high flux of oxygen at a low concentration. Haemoglobins seem to be present in low concentrations in many plants but nodulation dramatically increases the concentration in legumes. Succinate seems to be the carbon substrate of choice for rhizobia in nodules.
Haemoglobin in nodule
Structure of plant membranes and bacteroids
in nodule
The basic mechanisms of nitrogenase action are the same in symbiotic
and asymbiotic organisms.